ESCO-TDS1200Ⅱ IR Infrared Heating Temperature-Programmed Desorption Analyzer
 ESCO-TDS1200Ⅱ IR Infrared-Heated Thermal Desorption Spectrometer
ESCO-TDS1200Ⅱ IR Infrared-Heated Thermal Desorption SpectrometerFeatures
The ESCO-TDS1200Ⅱ IR infrared-heated thermal desorption analyzer is an analytical instrument that uses a quadrupole mass spectrometer (QMS) to observe in real time the molecules desorbed when a sample is programmatically heated by infrared radiation in an ultra-high vacuum environment (10-7Pa or lower).
Compared to thermal analysis instruments that heat samples in an atmospheric pressure environment (105Pa), this system enables detection in a low-background environment. It can detect atmospheric components (such as water, hydrogen, oxygen, nitrogen, and carbon dioxide) with high sensitivity, even though achieving low background levels is difficult in atmospheric pressure environments.
Furthermore, the system is designed so that only the sample is heated, preventing background noise from increasing even at high temperatures and enabling highly sensitive detection of desorbed molecules.
It allows for the identification and quantification of chemical species from molecules desorbed from the sample. Information regarding the adsorption and binding states of desorbed molecules, as well as their diffusion processes, can also be obtained.
It is ideal for measuring thin-film samples and thin-plate samples. The new ESCO-TDS1200Ⅱ IR is a revamped version of the conventional TDS1200 system and features a touch panel interface. It complies with CE marking standards.
[References]
●Edited by The Japan Society of Surface and Vacuum Science; Illustrated Handbook of Surface Analysis. Asakura Shoten, 2021, Chapter V: Others. Section 25: Desorption Analysis Methods. Item 1: Thermal Desorption Method (Shohei Ogura): 464-469.
●Norio Hirashita; Tsuneo Ajioka; Yasushi Hinanaga. Development of a New Thermal Desorption Gas Analyzer and Its Application to VLSI Material and Process Evaluation. Vacuum, 1991, 34.11: 813-819.
●Norio Hirashita; Taizo Uchiyama. Quantitative Analysis of Gases Emitted from Semiconductor Integrated Circuit Materials Using the Thermal Desorption Gas Analysis Method. Analytical Chemistry, 1994, 43.10: 757-764.
 Load Lock Chamber Configuration
Load Lock Chamber ConfigurationLoad Lock Chamber Configuration
A load lock chamber is essential for achieving high measurement efficiency (high throughput) and high sensitivity. Our load lock chamber and sample transfer mechanism allow only the sample to be quickly introduced into the ultra-high vacuum analysis chamber.
Without a load lock chamber, the analysis chamber must be vented to atmosphere every time a sample is exchanged. Once vented, a large amount of atmospheric components (especially moisture) are adsorbed inside the analysis chamber, requiring a long time to evacuate them completely.
 Image of Infrared Heating Method
Image of Infrared Heating MethodInfrared Heating Method
In thermal desorption analysis, it is crucial that the analysis chamber and its internal components do not heat up when the sample is heated. In TDS systems where the chamber itself heats up, it becomes difficult to distinguish between desorption gases from the sample and those from the chamber.
The TDS1200Ⅱ directly heats the sample using infrared radiation introduced via a quartz rod, minimizing increases in background levels even in high-temperature ranges and enabling highly sensitive measurements.
 Quadrupole Mass Spectrometer
Quadrupole Mass SpectrometerQuadrupole Mass Spectrometer (QMS) Configuration
To detect desorbed gases with high sensitivity, the QMS ionization chamber is positioned directly above the sample. By placing it directly above the sample, gases desorbed from the sample can reach the QMS ionization chamber directly, enabling the high-sensitivity detection of metals with high vapor pressure and organic molecules with large molecular weights.
Quantification of Desorbed Gases
Quantification of desorbed gases is possible using the data processing program. To perform this quantification, the mass spectrometer’s sensitivity must be calibrated periodically.
Sensitivity calibration using standard leaks requires preparing an equal number of expensive standard leaks for each gas type, making the calibration process time-consuming. Furthermore, when using standard leaks for toxic gases, strict safety and health management protocols are required.
Our current quantification program enables the quantification of desorbed gases more quickly, easily, and safely than methods using standard leaks. Simply by periodically measuring our NIST-traceable hydrogen standard samples, you can obtain highly accurate results. The sensitivity correction method we have developed can correct sensitivity for gases other than hydrogen as well, and it has been confirmed to agree well with quantitative results calibrated using standard leaks based on the national standards of the National Institute of Advanced Industrial Science and Technology (AIST).
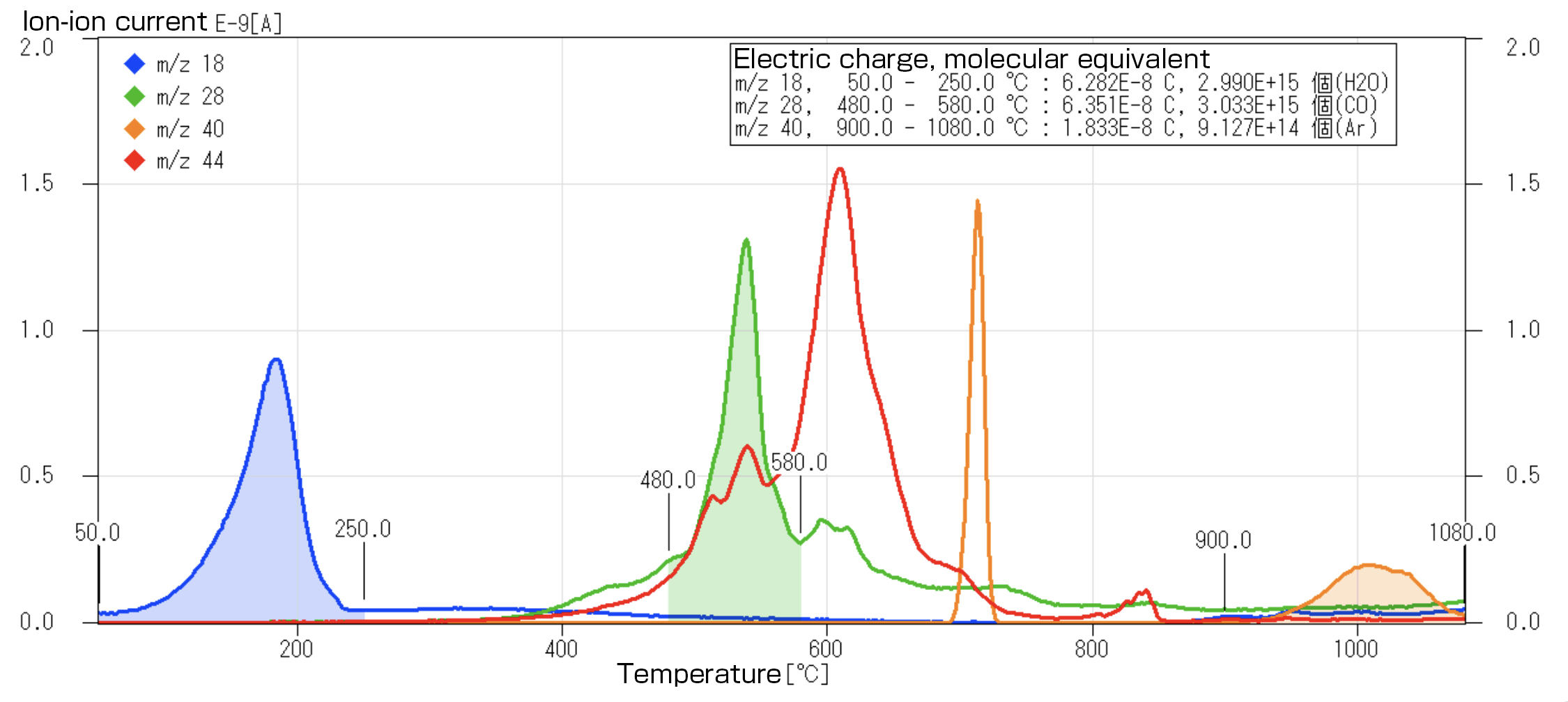
【References】
● Norio Hirashita; Mari Urano; Hajime Yoshida. Gas Release Measurement in the Field of Analysis. Journal of the Vacuum Society of Japan, 2014, 57.6: 214-218.
Technical Explanation / Thermal Desorption Method
This is reference material regarding the desorption models used in the analysis of the thermal desorption method and methods for determining activation energy.

*A PDF file will open.
Technical Explanation / Quantitative Analysis (Quantification Using a Thermal Desorption Analyzer)
This document is based on the report by Hirashita and Uchiyama in *BUNSEKI KAGAKU*, 43, 757 (1994).
The quantity of desorbed gas can be determined from the thermal desorption spectrum measured using a thermal desorption analyzer.
When the exhaust rate of the measurement chamber is sufficiently greater than the pressure change in the chamber caused by the desorbed gas, the change in the partial pressure of the desorbed gas is proportional to the amount of desorption per unit time (desorption rate).
Since ion current and partial pressure are proportional in a mass spectrometer, the ion current is ultimately proportional to the desorption rate, and the total desorption amount can be calculated from the area intensity obtained by integrating the ion current.
By determining the proportionality constant between area intensity and desorption amount using a Si sample injected with a known amount of H+, the hydrogen desorption amount can be determined from the area intensity at m/z 2 for various samples.
Furthermore, for molecules other than hydrogen, the proportionality constant for the target molecule can be calculated based on parameters such as the ionization ease, fragmentation factor, and transmission rate of both hydrogen and the target molecule. Using this proportionality constant, it is also possible to quantify molecules other than hydrogen.

TDS1200Ⅱ Thermal Desorption Analyzer [Optional]
 Switch-type valve section
Switch-type valve sectionGas purge for powder samples
This mechanism allows the load lock chamber to be gas-purged without dispersing fine powder samples.
| Purpose | ・Purging the load lock chamber containing powdered samples causes the samples to scatter. ・A switch-type valve is used in the purge line. ・Easy to operate, eliminating sample scattering. |
|---|---|
| Configuration | Slow-leak valve assembly (SUS piping, needle valve, etc.) |
| Applicable Models | ・TDS1200Ⅱ ・TDS1200 ・EMD-WA1000S/W ・EMD-WA1000S |
Transfer Vessel
This allows samples prepared in anaerobic environments, such as glove boxes, to be introduced into the load lock chamber without coming into contact with the atmosphere.

![]()

![]()

| Specifications | Air exchange rate = 1.5 MPa → 1.5 MPa (after 48 hours) |
|---|---|
| Configuration | ・Transfer vessel (available with pressure gauge) ・Load lock chamber for mounting transfer vessel |
| Applicable Options | ・TDS1200Ⅱ ・TDS1200 ・EMD-WA1000S/W ・EMD-WA1000S |
| Other | We also sell individual parts. Installation on a TDS requires modification of the load lock. |
Sample Stage Exchange Mechanism
Sample stages can be exchanged without exposing the main chamber to the atmosphere.
Transparent stages and SiC stages can be easily swapped depending on the heating conditions.
Even if the sample stage becomes contaminated due to sample sublimation, it can be replaced immediately, providing peace of mind.
Cooling Water Circulation Unit
Cooling water circulation is required to cool the infrared focusing mirror.
If the installation environment lacks a cooling water supply system, we recommend using a cooling water circulation unit in conjunction with the system.
You can choose from models ranging from basic cooling water circulation units to those equipped with various optional features.
When combined with a leak sensor, the system can automatically shut off the cooling water circulation unit and infrared heating upon detecting a leak.
 QMS Manifold
QMS ManifoldQMS Manifold
A space for installing the QMS analysis tube is provided between the main chamber and the turbo molecular pump.
This reduces metal deposition on the analysis tube and suppresses degradation of QMS sensitivity.
Additionally, installing a liquid nitrogen trap allows for more precise determination of hydrogen content in steel.


2kW heating source
Heating, which is typically limited to 1200°C, can be increased to 1400°C.
Consumables
